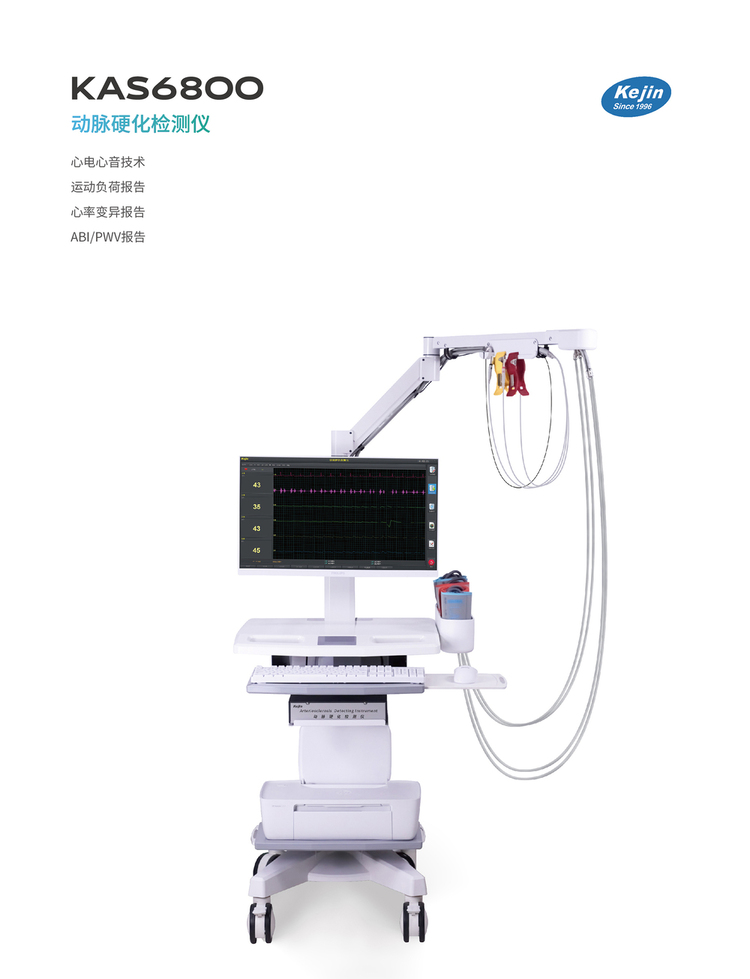
KAS6800 Arteriosclerosis Detector
KAS6800 Arteriosclerosis Detector
Product Parameters:
Product Name: Arteriosclerosis Detector
Model: KAS6800
Brand: Kejin
Medical/Home Use: Medical
Domestic/Imported: Domestic
Manufacturer: Nanjing Kejin
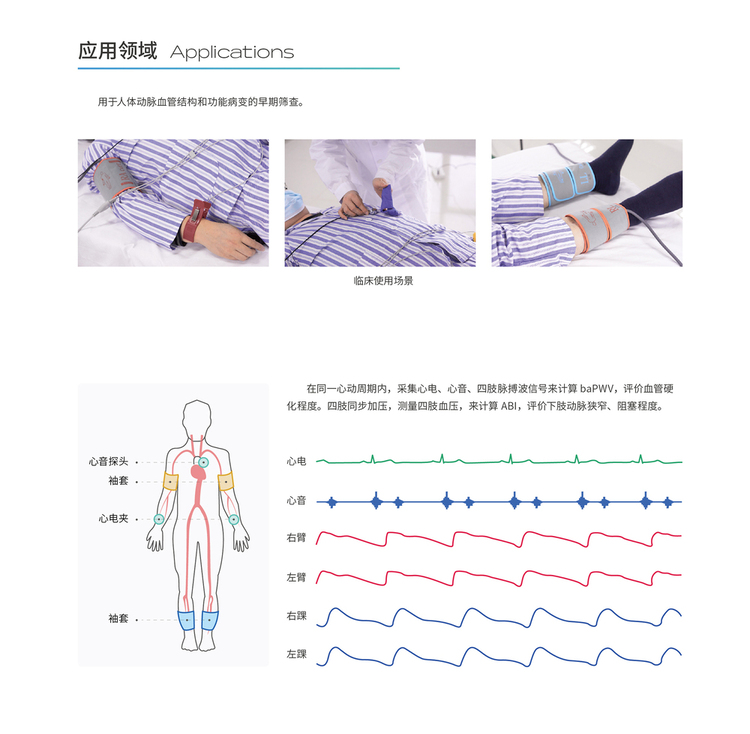
Scope of Application:
This product is used for early screening of human arterial structure and functional lesions.
Product Introduction:


Common Parameter Description:
ABI is the ratio of ankle systolic pressure to brachial systolic pressure. According to the results, the stenosis and obstruction of lower limb arteries can be judged. ABI is important for the diagnosis and postoperative observation of obstructive arteriosclerosis.
Pulse wave conduction velocity: When the heart contracts and blood is squeezed, the speed at which the pulse is transmitted from the heart to the end of the artery is mainly used to evaluate the degree of arterial wall sclerosis.
Applicable Population:
(1) Over 20 years old; diagnosed with hypertension (including borderline hypertension), hyperlipidemia, diabetes (including impaired fasting glucose and impaired glucose tolerance), coronary heart disease, and stroke;
(2) Those with a family history of early cardiovascular and cerebrovascular diseases, obesity, long-term smoking, high-fat diet, lack of physical exercise, and other high-risk factors for cardiovascular and cerebrovascular diseases;
(3) Those with long-term dizziness and discomfort, not yet clearly diagnosed; those with chest tightness or palpitations after activity or at rest, not yet clearly diagnosed;
(4) Health check-up individuals.
Note: The above description is excerpted from the product manual.
Medical Device Name: Arteriosclerosis Detector
Medical Device Manufacturer: Nanjing Kejin
Medical Device Registration Certificate No.: SuXieZhuan 20222071989
Medical Device Advertisement Approval No.: SuXieGuangShen (Wen) No. 251221-00212
Advice: Please read the product manual carefully or purchase and use under the guidance of medical staff. For contraindications or precautions, see the manual.